Flourishing 5K: Celebrating a Georgia CTSA Publications Milestone

At the end of 2025, the Georgia Clinical & Translational Science Alliance (Georgia CTSA) surpassed 5,000 publications supported since 2007, marking a major milestone in advancing clinical and translational research. This portfolio reflects not only sustained productivity but also the growing impact of research that informs clinical care, health policies, and improves health outcomes across Georgia and beyond.
Since its inception, the Georgia CTSA has supported research through a network of research resources, expert consultations, training programs, funding, and cross-institutional collaboration—creating a strong foundation for continued translational growth and impact.
This milestone was evaluated and highlighted by the Georgia CTSA’s Evaluation & Continuous Improvement program, which plays a central role in tracking research outcomes and assessing the impact of Georgia CTSA-supported work through ongoing analysis of publication productivity, collaboration patterns, and research influence. Publication output has been consistent since 2007, encompassing a wide range of clinical and translational research areas, including areas of high public interest, such as COVID-19, maternal health, and rural health.
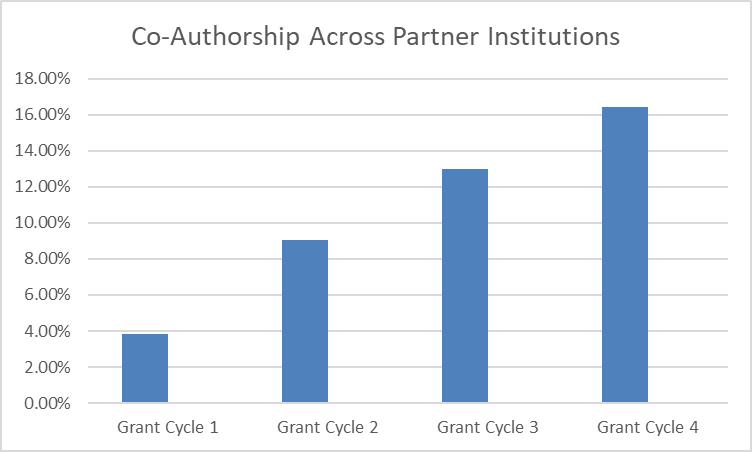
Co-authorship among Georgia CTSA institutions has increased from 3.9% in the first 5 years to 16.5% in the past 5 years, demonstrating a clear progression and strengthening collaboration across this multi-institutional consortium. As Eric J. Nehl, PhD, Director of Evaluation & Continuous Improvement, notes, “More than 5,000 research papers reflect the impact of collaborative science, advancing public health and accelerating translational discovery across Georgia. This milestone highlights the Georgia CTSA’s role in bringing researchers together to drive meaningful, lasting progress.”
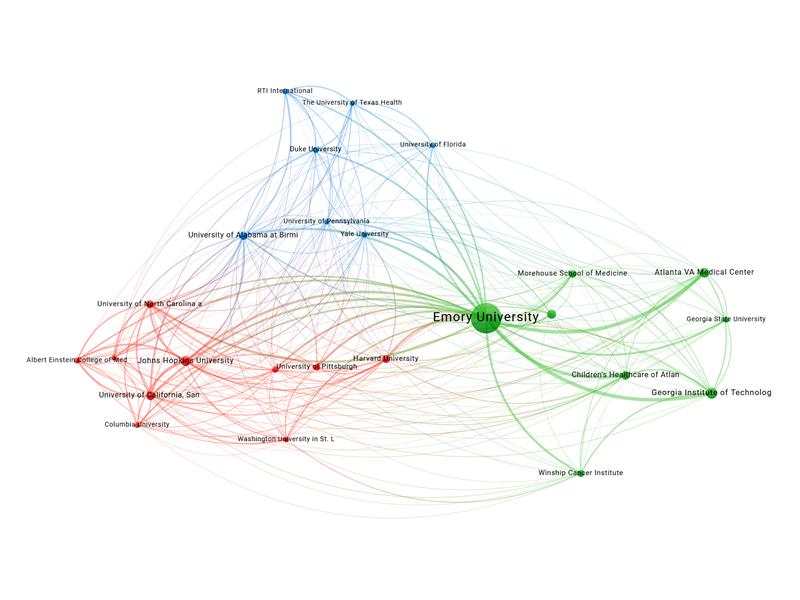
Co-Authorship Across Institutions
The influence of this work extends well beyond the number of publications. Georgia CTSA-supported publications have garnered more than 250,000 academic citations—more than twice the expected rate—and approximately 100,000 mentions in Altmetric (i.e., public/media) sources, reflecting strong engagement across both scientific and public audiences.
One example of this impact is seen in a 2024 publication in the New England Journal of Medicine, Omalizumab (Xolair) as a Treatment for Individuals with Multiple Food Allergies. Supported through the Georgia CTSA Pediatrics program, this work represents the successful translation of clinical research into meaningful advancement in treatment. The publication presenting this research, described as a life-changing innovation, has been referenced in more than 200 news stories, over 500 posts on X, several patent applications and policy documents, as well as 250 academic articles, demonstrating its reach across public, clinical, and scientific communities.
Nikki Llewellyn, PhD, Associate Director of Evaluation & Continuous Improvement, adds, “Examining how this portfolio of publications has been used beyond academia sheds light on the translational impact of the Georgia CTSA, from bench to bedside. It allows us to measure and describe the progress that has been made across science, medicine, and public health.”
As the portfolio continues to grow, the first 5,000 publications represent a strong foundation for continued expansion and impact in the years ahead.